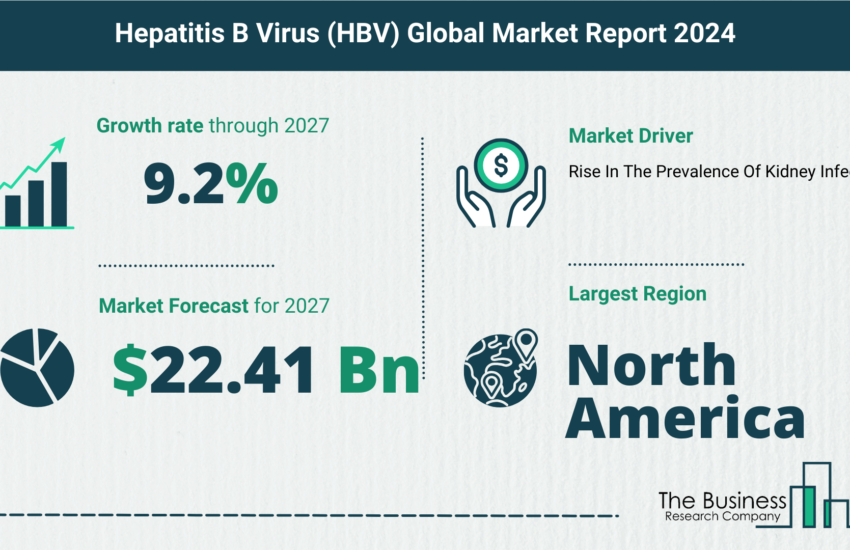
Research Initiatives And Funding And Demand For New Medicines For Drug-Resistant Infections In The Anti-Infective Drugs Industry
Anti-infective drug manufacturers are investing in programs to develop drugs to treat drug resistant infections. Antimicrobial resistance is a serious concern to global public health as it results in long duration of illness, the requirement of additional tests and use of more expensive drugs. Antimicrobial resistance occurs when microorganisms such as bacteria, viruses, parasites and fungi change and adapt to drugs when they are exposed to antimicrobial drugs such as antibiotics, antivirals, antifungals, anthelmintics and others. According to the World Health Organization (WHO), globally 4,80,000 people are developing multi-drug resistant tuberculosis (MDR-TB) each year, and drug resistance is starting to complicate the treatment of malaria and HIV as well. Due to rising number of drug-resistant bacteria, companies are investing in developing new drug candidates to treat drug-resistant infections. For instance, in 2014, Cubist Pharmaceuticals launched a new antibiotic Zerbaxa (ceftolozane/tazobactam) for complicated intra-abdominal infections, complicated urinary tract infections and also for infections of the kidney (pyelonephritis). Similarly, in June 2018, plazomicin was approved by the US Food and Drug Administration (FDA) for the treatment of complicated, drug-resistant urinary tract infections
Read More On The Global Anti-Infective Drugs Market Report At
https://www.thebusinessresearchcompany.com/report/anti-infective-drugs-market
Pharmaceutical companies and federal governments are increasingly working together in partnerships and collaborations to provide funding and implement incentive programs for the research and development (R&D) of anti-infective drugs. These partnerships provide financial and technical assistance across different clinical development phases to pharmaceutical companies. Various programs such as the Innovative Medicines Initiative’s (IMI’s) New Drugs for Bad Bugs (ND4BB) program, Joint Programming Initiative on Antimicrobial Resistance (JPIAMR), and Biomedical Advanced Research and Development Authority’s (BARDA) Broad Spectrum Antimicrobials Program have been implemented for the development of novel antibiotics by focusing on R&D gaps. Similarly, Global Antibiotic Research and Development Partnership (GARDP) was set up by the World Health Organization (WHO) and the Drugs for Neglected Diseases Initiative (DNDi) in May 2016. This R&D initiative focuses on development of new or improved antibiotic treatments
Request A Free Sample Of The Anti-Infective Drugs Market Report At
https://www.thebusinessresearchcompany.com/sample.aspx?id=2348&type=smp
The anti-infective drugs industry consists of sales of anti-infective drugs and related services by entities (organizations, sole traders and partnerships) that produce anti-infective drugs to treat microbial infections. This industry includes establishments that produce antibiotics to treat bacterial infections, anti-viral drugs to treat viral infections, anti-fungal drugs to treat fungal infections, anti- helminthic drugs to kill internal parasites and antiprotozoal agents.
The pharmaceutical drugs market, of which the anti-infective drugs market is a segment, develops drugs to diagnose, cure, treat or prevent diseases. Pharmaceutical drugs are subjected to various laws and regulations that deal with patenting, testing, safety, efficacy and marketing.
Read More On The Global Pharmaceutical Drugs Market Report At
https://www.thebusinessresearchcompany.com/report/pharmaceutical-drugs-market
The anti-infective drugs market report covers market characteristics, size and growth, segmentation, regional and country breakdowns, competitive landscape, market shares, trends and strategies for this market. It traces the market’s historic and forecast growth by geography. It places the market within the context of the wider pharmaceuticals market, and compares it with other markets.